IASR 43(11), 2022【THE TOPIC OF THIS MONTH】Influenza 2021/22 season, Japan
PDF download(PDF:5,463KB)
The topic of This Month Vol.43 No.11(No. 513)![]()
Influenza 2021/22 season, Japan
(IASR Vol. 43 p243-246: November 2022)
In the 2021/22 influenza season (week 36, 2021, to week 35, 2022), the number of reported influenza cases did not increase during the usual epidemic period (around week 45 to week 19 of the following year). Multiple other indicators also remained at remarkably low levels, suggesting that there was no apparent epidemic, as in the previous season. However, the number of reported cases continues to exceed that of the previous two seasons from around week 27 of 2022, raising concerns about a resurgence in influenza activity.
Epidemiology of the 2021/22 influenza season (as at 5 October 2022)
Under the National Epidemiological Surveillance of Infectious Diseases (NESID) system, the number of influenza cases is reported every week from approximately 5,000 influenza sentinel sites (approximately 3,000 pediatric and 2,000 internal medicine facilities). The maximum weekly number of reported influenza cases per sentinel in the 2021/22 season was 184 (0.038 per sentinel site/week) in week 29 of 2022, and, as in the previous season, there was no week in which the number of reports per sentinel exceeded 1.00, which is the indicator for the nationwide start of the influenza season (Fig. 1). Notably, the number of reports has continued to exceed that of the previous season since around week 27 of 2022.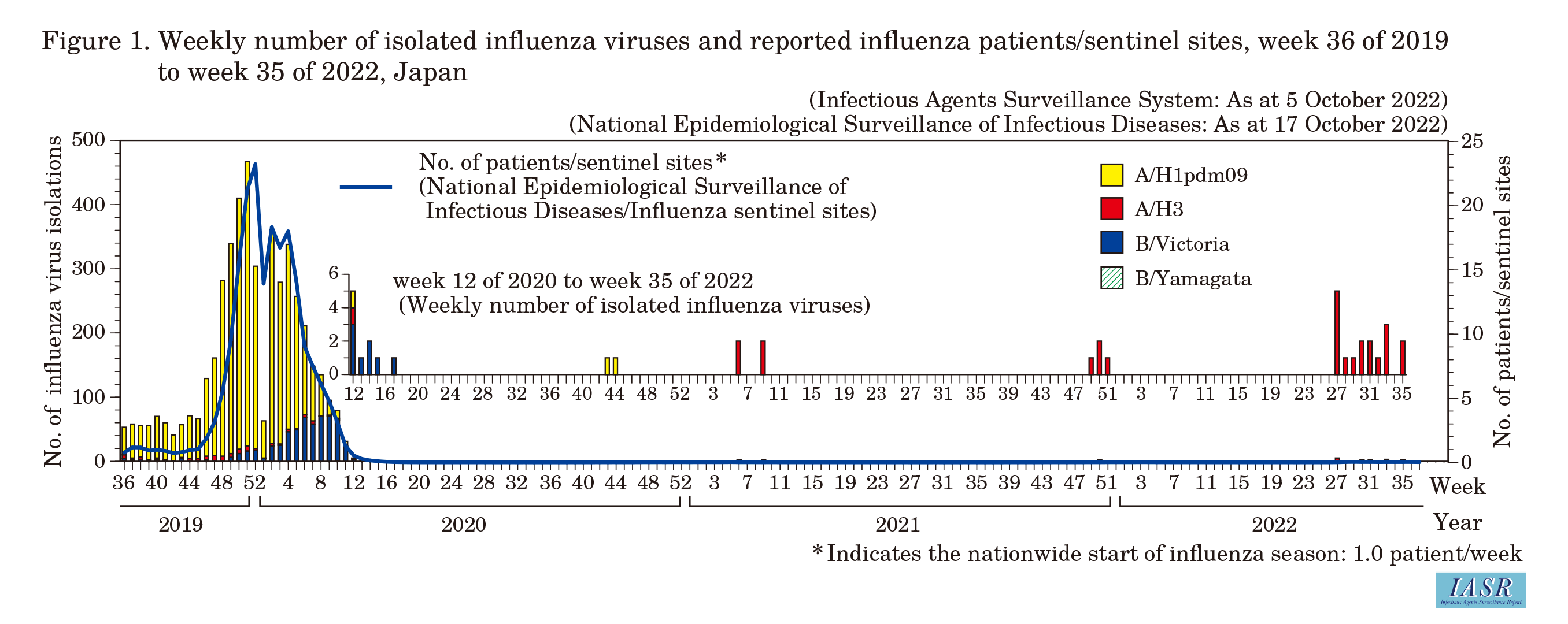
Based on the reported sentinel surveillance data, the estimated cumulative number of medically-attended influenza patients was approximately 3,000 (week 36 of 2021 to week 17 of 2022); this estimate was below the estimated number of patients for the same period in the previous season (approximately 14,000), which was markedly lower than the number of patients estimated for the same period in the prior season (approximately 7,289,000).
According to hospitalized influenza patient surveillance (data from approximately 500 designated sentinel hospitals with at least 300 beds), the total number of hospitalized patients was 44, even lower than the previous season (278 cases) that was a substantial decrease from those observed in usual years.
One case of acute encephalitis (including encephalopathy), a category V notifiable infectious disease, was reported as influenza encephalopathy. As in the previous season (0 cases), the number of cases was considerably lower than that of an average year.
Based on these data, as in the previous season, it was concluded that there was no influenza epidemic in the 2021/22 season.
Isolation/detection of influenza virus in the 2021/22 season (as at 17 October 2022)
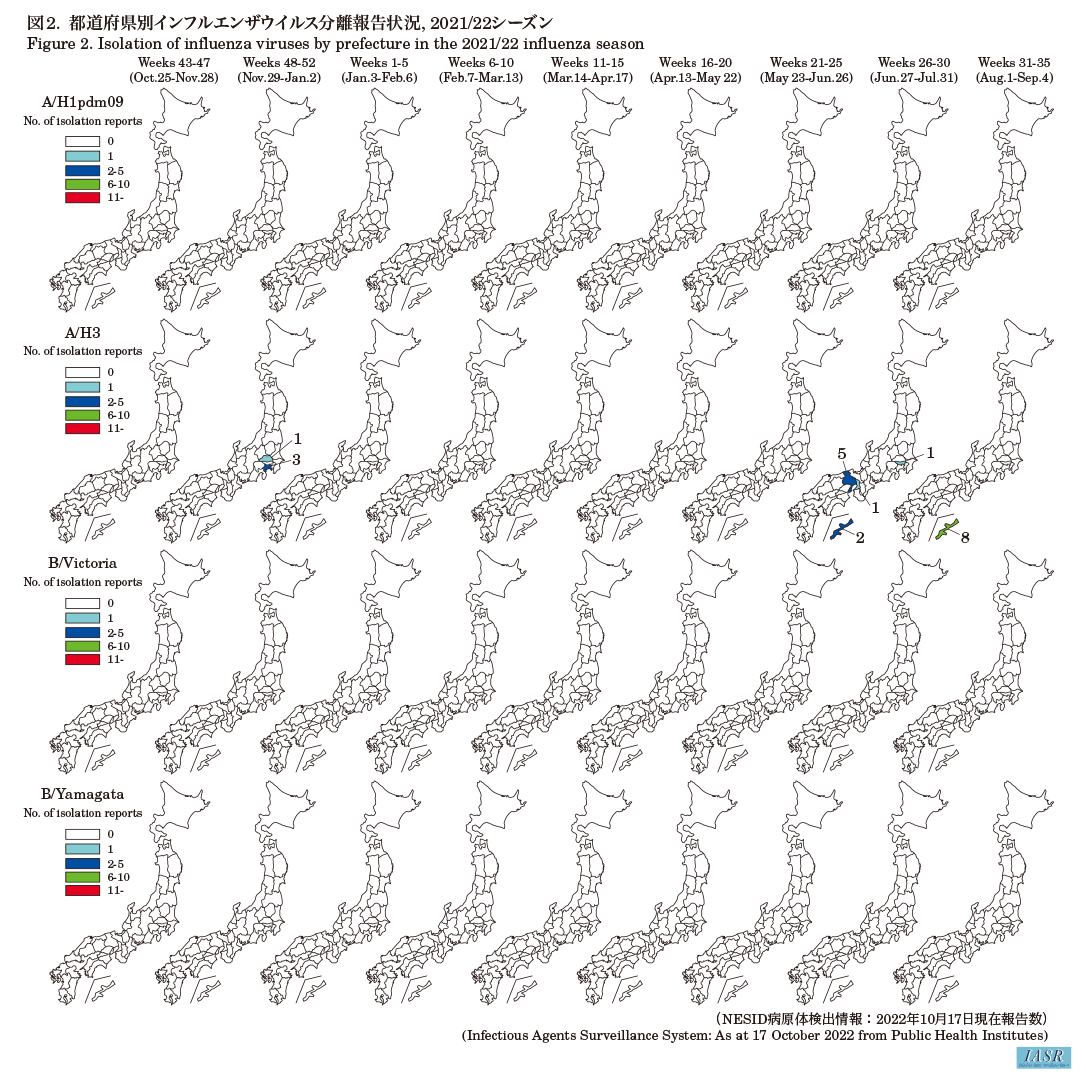
Prefectural and municipal public health institutes reported the isolation or detection of 45 influenza viruses in the 2021/22 season (Table 1). Among them, 17 were reported from influenza sentinel sites and 28 from non-sentinel sites (Table 2). By type/subtype, three were influenza subtype A/H1pdm09 and 42 were subtype A/H3, and there were no reports of type B virus (Victoria or Yamagata lineage) or type C virus (Table 2). The number of virus isolates (not including reports based on viral gene detection) during the usual influenza epidemic period was four A/H3 subtypes; however, 17 A/H3 subtypes have been isolated and reported since week 25 of 2022 (Fig. 1 & Fig. 2).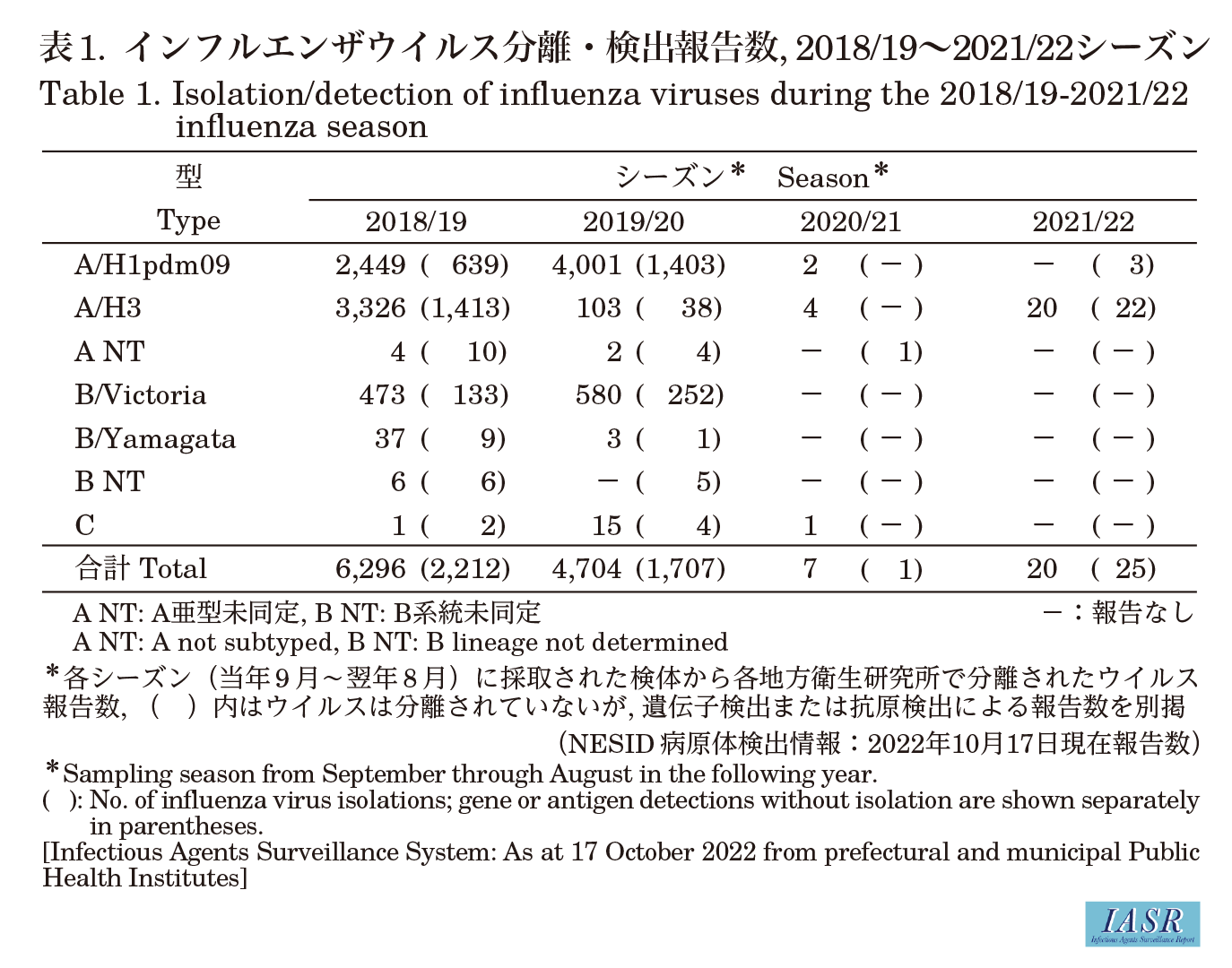

Genetic and antigenic characteristics of 2021/22 isolates
Genetic analyses and antigenic analyses using ferret antiserum (see p.247 of this issue) were conducted for domestic and Asian isolates at the National Institute of Infectious Diseases (NIID). The subtype A/H1pdm09 strains belonged to genetic subclade 6B.1A.5a.1 or 6B.1A.5a.2 according to the phylogenetic tree analysis of the hemagglutinin (HA) gene. Antigenic analysis revealed that the isolates belonging to subclade 6B.1A.5a.2 reacted well with ferret antiserum immunized with the “egg-isolated” A/Victoria/2570/ 2019 (6B.1A.5a.2), the World Health Organization (WHO)-recommended vaccine strain for 2020/21, whereas the isolates belonging to subclade 6B.1A.5a.1 did not react well with the antiserum. Antigenic analysis using post-vaccinated human sera showed that isolates belonging to 6B.1A.5a.2 reacted well with the sera, and viruses belonging to 6B.1A.5a.1 also reacted generally well. For subtype A/H3 viruses, HA gene phylogenetic tree analysis showed that all viruses analyzed belonged to subclade 3C.2a1b.2a.2. Antigenicity analysis showed that all viruses analyzed reacted well with ferret sera against viruses belonging to 3C.2a1b.2a.2, but not with human sera vaccinated with the WHO-recommended vaccine strain [A/Cambodia/e0826360/20 (3C.2a1b.2a.1)]. For B/Victoria viruses, HA gene analysis showed that most viruses belonged to subclade 1A.3a.2. Antigenic analysis showed that none of the tested strains reacted well with ferret sera against the WHO-recommended vaccine strain, B/Washington/02/2019 (1A.3), and reacted well with ferret sera against viruses belonging to 1A.3a.2. The reactivity of viruses belonging to 1A.3a.1 and 1A.3a.2 with post-vaccinated human sera tended to be low. No strains of the B/Yamagata lineage were analyzed.
Resistance to antivirals among 2021/22 isolates (see p.247 of this issue)
In Japan, four neuraminidase (NA) inhibitors (Oseltamivir, Zanamivir, Peramivir, Laninamivir), a cap-dependent endonuclease inhibitor (Baloxavir), and an M2 inhibitor (Amantadine) are used as anti-influenza drugs. A strain of A/H1pdm09 subtype virus isolated in Japan during the 2021/22 season was not resistant to NA inhibitors or Baloxavir, but was Amantadine-resistant. Twenty-three strains of A/H3 subtype viruses isolated in Japan and overseas were not resistant to NA inhibitors or Baloxavir, but were resistant to Amantadine. Five strains of type B virus isolated overseas were not resistant to NA inhibitors or Baloxavir.
Seroprevalence in the Japanese population before the 2021/22 season
The age-group specific prevalence of antibodies (HI antibody-positive, titer ≥ 1:40) against domestic influenza vaccine strains for the 2021/2022 season was investigated by seroprevalence surveys, using serum samples (3,448 persons) collected between July and September 2021, conducted under the National Epidemiological Surveillance of Vaccine-Preventable Diseases program. The seroprevalence for A/Victoria/1/2020 (IVR-217) [subtype A/H1pdm09 vaccine strain] was highest at 38% in the 10-14-year-olds and was 10-18% in the 30-59-year-olds. The age group with the highest seroprevalence for A/Tasmania/503/2020 (IVR-221), [subtype A/H3N2], was the 20-24-year-olds (54%), while the other age groups were 41% or less. Seroprevalence for B/Phuket/3073/2013 [B/Yamagata lineage vaccine strain] was greater than 60% in all age groups among those aged 20-39 years, but 18% or less in those aged 70 years and older. Seroprevalence for the B/Victoria strain B/Victoria/705/2018 (BVR-11) [B/Victoria lineage vaccine strain] was 40% or greater in all age groups among those aged 40-54 years, higher than that in the other age groups.
Influenza vaccine
In the 2021/22 season, approximately 28,670,000 vial-equivalent doses (estimated on the assumption that 1 vial contained 1 mL, 0.5 mL is used per vaccination, likewise hereafter) of tetravalent vaccine (containing two subtype A and two type B strains) were produced, and an estimated 25,970,000 vials were used.
For the 2022/23 season, A/H1pdm09 subtype A/Victoria/1/2020 (IVR-217), A/H3 subtype A/Darwin/9/2021 (SAN-010), B/Yamagata lineage B/Phuket/3073/2013, and B/Victoria lineage B/Austria/1359417/2021 (BVR-26) were selected as the vaccine strains (https://www.niid.go.jp/niid/ja/flu-m/flu-iasrd/11145-507s03.html).
With influenza epidemics being seen worldwide (see p.255 of this issue), there is concern of a domestic epidemic, and the production of 35,210,000 vial-equivalent doses of vaccine is expected for the next season, the largest number ever based on recorded data.
Avian and swine influenza virus outbreaks and human cases
Human cases of A(H5N1) virus infection have been reported mainly from Asia and Africa since 2003; however, for the first time, a human case was reported from the United Kingdom in January 2022 and from the United States in April 2022, resulting in a total of 865 cases (456 deaths). Since 2013, 81 human cases of A(H5N6) virus infection have been confirmed, mainly in China, 36 of which occurred since September 2021. Since 2013, 1,568 human cases of A(H7N9) virus infection (616 deaths) have been confirmed, but no cases have been reported after March 2019. Twenty-one human cases of A(H9N2) virus infection have been confirmed in China since September 2021. One human case of A(H3N8) virus infection was reported in April and another in May 2022, but human-to-human transmission has not been confirmed.
Since swine are infected not only with influenza viruses of mammalian origin but also with those of avian origin, viral gene reassortment can occur in swine, resulting in the emergence of new viruses. Since August 2021, human cases of A(H1N1) variant (v), A(H1N2)v, A(H3N2)v, and A/H1v (NA type unknown) infection have been confirmed in North America, China, and Europe. In Japan, since 2009, A(H1N2) viruses and A(H3N2) viruses that have genetically reassorted with A(H1N1) pdm09 viruses have been detected in swine, indicating that close monitoring is necessary (see p.257 of this issue).
In addition, since the highly pathogenic avian influenza virus A(H5N1) has been detected in wild birds and animals in Japan, attention should be paid to changes in the epidemiology of the virus (see p.259 of this issue).
Conclusion
As in the 2020/21 season, it was concluded that there was no influenza epidemic in the 2021/22 season in Japan. On the other hand, starting around week 27 of 2022, the number of reported cases has continuously exceeded that of the same period in the past two seasons, raising concerns of a future influenza epidemic. In such a case, a simultaneous epidemic with novel coronavirus infection (COVID-19) is possible, with concerns of a strained medical care system. Public health measures, such as the vaccination of high-risk groups, should be implemented in preparation for increased influenza activity. In addition, comprehensive influenza surveillance systems should be strengthened, including monitoring of influenza activity through the NESID system, monitoring of circulating viruses through genetic analysis, antigenic analysis, and drug resistance analysis based on the NESID infectious agents surveillance system, as well as seroprevalence surveys of the population.
Note: For IASR reporting, influenza nomenclature is based on the virus information available. Influenza viruses are classified by type, subtype, lineage, and strain based on hemagglutination (HA), neuraminidase (NA), and other information including nucleotide sequences:
- When both HA and NA typing have been performed, the names are listed fully [e.g., A(H1N1)pdm09, A(H3N2), A(H5N1)].
- When NA typing has not been performed, only HA information is listed (e.g., A/H1pdm09, A/H3).
- The strain name is represented by the location of isolation; in the case of isolation in Japan, the location is written in Japanese kanji, and in the case of isolation in foreign countries, in English.
- To distinguish swine influenza viruses recovered from humans from seasonal influenza viruses, the term “variant virus” is used with the letter “v” added after the subtype name [e.g., A(H3N2)v].

