Current National and Global Status of Severe Fever with Thrombocytopenia Syndrome (SFTS)
August 1, 2024
Revised September 18, 2025
National Institute of Infectious Diseases, Japan Institute for Health Security
Department of Virology I
Department of Veterinary Science
Department of Medical Entomology
Center for Public Health Action in Applied Epidemiology
Department of Infectious Disease Surveillance
Center for Emergency Preparedness and Response
This document is intended for the timely sharing of information. Its contents and perspectives may change as the situation evolves. Please refer to the most recent updates for the latest information.
Table of Contents
- Overview
- Virological Findings
- Tick Vectors of SFTS virus
- Global situation
- Domestic Situation
- Domestic Countermeasures
- Risk Assessment
- Future Measures and Recommendations
1.Overview
Severe fever with thrombocytopenia syndrome (SFTS) is a tick-borne infectious disease caused by the SFTS virus (SFTSV), classified in the family Phenuiviridae, genus Bandavirus. The virus was first reported by Chinese researchers in 2011.
SFTS manifests with symptoms such as fever, fatigue, and headache following an incubation period of 6–14 days. Other common symptoms include lymphadenopathy and gastrointestinal symptoms such as anorexia, nausea, vomiting, diarrhea, and abdominal pain. Complications can include shock, acute respiratory distress syndrome, altered consciousness, renal dysfunction, myocardial dysfunction, disseminated intravascular coagulation (DIC), and hemophagocytic syndrome. Laboratory findings often demonstrate leukopenia, thrombocytopenia, and elevated hepatic transaminases. The case fatality rate (CFR) in Japan has been reported at 27%, while there are reports indicating 10% in China (Kobayashi et al., 2020).
Case management was previously limited to supportive care, as mild cases typically resolve spontaneously. However, on May 24, 2024, Second Committee on Drugs, Pharmaceutical Affairs Division II, Pharmaceutical Affairs Council, Ministry of Health, Labour and Welfare (MHLW), approved favipiravir for treatment of SFTS. Official approval to this additional indication against SFTS was granted on June 24, 2024.
In Japan, the first case of SFTS without overseas travel history was reported in January 2013. On March 4, 2013, SFTS (limited to cases caused by the SFTSV of the genus Phlebovirus*) was designated as a Category 4 Infectious Disease in the Infectious Disease Surveillance System under the Act on the Prevention of Infectious Diseases and Medical Care for Patients with Infectious Diseases (Infectious Diseases Control Law). The SFTSV of the genus Phlebovirus is also classified as a Class III pathogen. Since then, SFTS has been designated as a notifiable disease under the national infectious disease surveillance system. As of July 31, 2025, a total of 1,185 cases has been reported.
SFTS is primarily transmitted via tick bites, particularly from Haemaphysalis longicornis. However, transmission has been reported among veterinary professionals after contact with infected animals, as well as through direct contact with patient blood or body fluids. In March 2024, Japan has documented its first case of human-to-human transmission.
Until recently, cases were geographically concentrated in western Japan, with presumed infection areas in Chubu region and areas westward. However, in July 2025, Kanagawa Prefecture reported the first case presumed to have been infected the Kanto region. In August 2025 by a case presumed to have been infected in Sapporo, Hokkaido. These findings suggest that the risk of infection now exists nationwide.
*:This term is used in the notification criteria under the Infectious Diseases Control Law and may differ from the current academic classifications.
2. Virological Findings
The SFTSV (SFTSV) was isolated in 2009 from a patient with a febrile illness accompanied by severe thrombocytopenia and leukopenia in China. SFTSV, also known as Bandavirus dabieense, belongs to the Phenuiviridae family, genus Bandavirus (Yu XJ, et al., 2011). The viral particles are spherical, approximately 100 nm in diameter, possess a lipid bilayer envelope structure. The virus contains four viral proteins (Gn, Gc, N, RdRp) and a three-segmented negative-strand RNA genome; The L segment, 6,255 nucleotides long, encodes the RNA-dependent RNA polymerase (RdRp), the M segment, 3,336 nucleotides long, encodes the envelope proteins (Gn/Gc), and the S segment, 1,681 nucleotides, encodes the nucleocapsid protein (N) and non-structural protein (NSs). Due to its biochemical characteristics of enveloped viruses possessing RNA as their genome, SFTSV is easily inactivated by heat, ultraviolet light, detergents, 70% ethanol, etc.
SFTSV is designated as a Class III pathogen under Japan's Infectious Diseases Control Law, as it causes SFTS, a hemorrhagic fever-like disease with high CFR in humans. At the National Institute of Infectious Diseases, it is classified as Biosafety Level 3 (BSL-3) under the National Institute of Infectious Diseases (NIID) Safety Management Regulations for Pathogens and Toxins (MHLW, 2023).
High viral load is detected in blood samples of patients with SFTS. The virus targets monocytes, macrophages, and B cells differentiating into plasmacytoid cells in humans. Viral replication in these target cells leads to disruption of immune responses, contributing to the manifestation of the disease. In vitro, SFTSV replicates efficiently in Vero cells (derived from African green monkey kidney) and Huh7 cells (derived from human hepatocytes), but exhibits weak cytopathic effects. In animal models, cats and aged ferrets inoculated with SFTSV are known to exhibit a fatal course similar to human SFTS. Conversely, infection is fatal only in immunodeficient and nude mice, with no clinical signs observed in immunocompetent mice after inoculation.
Genome sequences of patient-derived and isolated viruses have revealed over 90% sequence identity across the genome, with the relatively low conservation of the M segment, indicating this virus has relatively low mutation rates compared to other RNA viruses. Detailed molecular phylogenetic analysis of isolates from China and South Korea indicated that viral strains can be classified into two major lineages: those prevalent in China and in Japan. Reassortants (genetic reassemblies) with swapped segmented genomes in the third segment exist, although rare. Genome lengths differ among the virus group, but no strains with markedly distinct antigenic or pathogenic properties have been reported. The characteristics of SFTSVs isolated in wild or domestic animals cannot be distinguished from those isolated from patients.
Laboratory diagnosis of SFTS in the acute phase involves viral gene detection by RT-PCR from blood, saliva, urine, etc., antigen detection through cytopathology or histopathology, and virus isolation. In the convalescent phase, diagnosis can be made by detecting an increase in SFTSV-specific antibody titers using paired sera. Cross-reactivity with other Bandavirus genus viruses requires caution. For details, refer to the pathogen detection manual available on the National Institute of Infectious Diseases website (Infectious Disease Information Site) (National Institute of Infectious Diseases, 2024).
3.Tick Vectors of SFTS virus
SFTSV is transmitted to humans and other hosts through bites of infected ticks.
Experimental studies have confirmed two competent tick species, the longhorned tick (Haemaphysalis longicornis) and the yellow tick (Haemaphysalis flava), to transmit SFTSV. In Japan, the seasonal distribution of SFTS cases, most frequent from spring through autumn, corresponds to the peak activity of H. longicornis. However, sporadic cases during winter suggest that other tick species, such as the H. flava, whose nymphs and adults remain active in winter, may also contribute to transmission. Amblyomma testudinarium has been found attached to SFTS patients and its role as a vector is suspected, but not scientifically confirmed.
Among ticks inhabiting Japan, SFTSV has been detected not only in H. longicornis and H. flava but also in H. kitaokai, H. megaspinosa, and the A. testudinarium. Ticks possess a specialized digestive mechanism known as endophagous digestion, and may retain nucleic acid molecules from their blood-feeding hosts for prolonged periods. Therefore, detection of viral genes within the tick does not necessarily indicate that the species is involved in SFTSV transmission, as the tick may have ingested blood from a viremic host.
4.Global situation
1.East Asia
The world's first cases of SFTS were documented in China in 2011, following investigations into unexplained febrile illnesses occurring between 2008 and 2009 (Yu XJ. et al., 2011; Xu B. et al., 2011). Subsequently, SFTSV was retrospectively identified in cases as early as 2006 (Liu Y. et al., 2012). A 2018 report suggested possible SFTS cases as early as 1996, involving 14 cases including a family in Jiangsu Province, China, and treating physicians who experienced unexplained fever and thrombocytopenia. Retrospective serological testing using preserved serum samples from 6 of these cases reported that all 6 were positive for SFTSV antibodies (IgM positive by ELISA; one was also IgG-positive by indirect fluorescent antibody assay). 4 of these cases remained IgG-positive with titers ranging from 80 to 640-fold in serum samples collected in 2010, 14 years after onset. However, SFTSV genetic material was not detected in any of the samples (Hu J. et al., 2018).
In China, reporting of SFTS to China Information System for Diseases Control and Prevention has required as equivalent to a class B statutory infectious disease since 2010, although not designated as a statutory infectious disease under the Law on the Prevention and Control of Infectious Diseases. Unlike other statutory infectious diseases, the data are not regularly summarized but are analyzed and published. From 2010 to 2023, 27,447 cases and 1,326 deaths (CFR: 4.83%) were reported from 27 provinces (including autonomous regions and municipalities directly under the central government). During the period, the number of reported cases increased, while the CFR decreased from 12.68% in 2010 to 3.77% in 2023. Mortality exhibited a unimodal pattern, peaking annually from May to July, with 83.26% of deaths concentrated between April and August (Yue Y. et al., 2024a). The majority of cases were concentrated in seven provinces: Shandong, Henan, Anhui, Hubei, Liaoning, Zhejiang, and Jiangsu. Over 95% of cases were aged 40 years or older, and 85% were agricultural workers (China Centers for Disease Control (CDC) Bulletin, 2024). A report summarizing cases from 2010 to 2021 indicated a national incidence rate of 0.11 cases per 100,000 population during this period. However, it highlighted significant regional variation, with some areas in southern Henan Province exceeding 10 cases per 100,000 population. While males accounted for 52.72% of cases, the incidence rate was slightly higher among females at 0.12 cases per 100,000 population compared to 0.10 cases per 100,000 for males. Furthermore, the age distribution showed a tendency toward older age groups: 34.53% were in their 60s, 34.22% in 70s, 15.52% in 50s, and 11% were aged 80 or older (Yue Y. et al., 2024b).
In Taiwan, the first SFTS case was confirmed in November 2019, in a man in his 70s from the northern region with no recent overseas travel history but frequently visits to mountainous areas (Peng SH. et al., 2020). In May 2022, the Taiwan CDC reported a second case involving a forestry worker in eastern Taiwan (Taiwan CDC, 2023).
In South Korea, the first case was a fatality from SFTS in 2012, reported the following year (Kim KH. et al., 2013). From 2017 to 2020, annual SFTS case reports ranged from 200 to 250, and from 150 to 200 since 2021. In 2024, 170 cases (including 167 laboratory-confirmed) and 26 deaths (CFR 15.3%) were reported. Most cases in 2024 were reported between May and October. Those aged 50 and older accounted for 94.1% of cases, and males comprised 57.1%. Geographically, cases were concentrated in Gangwon, Gyeonggi, and North Gyeongsang provinces in northern and eastern South Korea. The main sources of exposure were agricultural work (51.8%) and individual outdoor activities (20%) (Korea Disease Control and Prevention Agency, 2025).
Cases of SFTS patients occurring in 2008, 2010, and 2012 were reported in 2018, identifying the 2008 case being the earliest documented SFTS case in South Korea (Kim KH. et al., 2018, Kim YR. et al., 2018).
Furthermore, in 2009, a 57-year-old male from North Korea developed acute thrombocytopenia, leukopenia, and multiple organ dysfunction while temporarily working in Dubai, United Arab Emirates. Although pathogen diagnosis was not performed, clinical symptoms suggested a possible SFTS infection case originating in North Korea (Denic S. et al, 2011).
Regarding risk factors for SFTS mortality, China reported that age, altered consciousness, and elevated LDH and CK levels were significantly associated with fatal outcome. A meta-analysis based on reports from China, South Korea, and Japan analyzed that age, viral load, thrombocytopenia, elevated liver enzymes, low albumin, and prolonged APTT were significantly associated with fatal outcome (Liu W. et al., 2013; Chen Y. et al., 2017).
2.Southeast Asia
Reports of SFTS cases, including retrospective diagnoses, have been documented in Southeast Asia in addition to East Asia.
In Thailand, PCR testing for SFTSV using stored specimens from patients hospitalized in Bangkok and surrounding areas between October 2018 and March 2021 identified three positive cases, occurring in November 2019, May 2020, and October 2020 Genome sequencing indicated that all three strains were closely related to SFTSV genotypes previously detected in China between 2012 and 2017. None of the patients had traveled overseas within three weeks prior to onset, suggesting infection occurred in the Bangkok area (Rattanakomol P. et al., 2022).
In Vietnam, PCR testing for SFTSV of serum samples from 80 febrile patients hospitalized at a hospital in Hue City, central Vietnam, between October 2017 and March 2018, detected two positive cases: a man and a woman in their 20s with no travel history to China, South Korea, or Japan. One of these cases also tested positive for IgM antibodies. Both cases presented with fever, headache, thrombocytopenia, leukopenia, and elevated serum liver enzymes, consistent with SFTS clinical findings (Tran XC. et al., 2019).
In Myanmar, PCR testing of 152 suspected tick-borne disease as Scrub typhus patients identified 5 SFTS cases. Genome analysis revealed that four strains were similar to those reported from Vietnam, while the remaining strain differed by only a single nucleotide (Win AM. et al., 2020).
To date, no confirmed SFTS cases have been reported outside East and Southeast Asia.
5.Domestic Situation
1. Human cases
SFTS was designated as a Category 4 Infectious Disease, a notifiable disease, under the Infectious Diseases Surveillance System based on the Infectious Diseases Control Law on March 4, 2013. Physicians who diagnose patients or asymptomatic carriers, or who perform postmortem examinations on deceased individuals suspected or confirmed to have died from infection, must immediately report to the nearest public health center.
From March 4, 2013, to July 31, 2025 (provisional data as of July 31), 1,185 cases, including eight cases that developed symptoms before the reporting system began, have been reported. Four cases that developed symptoms before reporting began are outside the surveillance system but are not included. Of these, 126 cases (10.6%) were fatal at the time of reporting; however, it should be noted that cases that died after reporting may not be included. Case reports consistently peak between May and October and have been recorded across 33 prefectures, predominantly in western Japan. In 2025, Akita, Kanagawa, and Gifu prefectures reported their first cases, with Kanagawa and Gifu prefectures being added to the list of the estimated region of infection (Figures 2 and 3). Although occurring after the current reporting period, patients with presumed infections in Hokkaido and Ibaraki Prefecture, neither of which had previous cases, were also reported in August (Ibaraki Prefecture, 2025; Sapporo City, 2025).
Female cases were 598 cases (50.5%), with a male-to-female ratio of approximately 1:1. The majority of patients were aged 60 years or older (Figure 1, Table 1). Twelve cases involved veterinary practitioners presumed to have been infected during medical treatment for animals, including one fatality.
Furthermore, although not reported in the surveillance system, a 2021 retrospective study of blood samples from patients with fever of unknown origin reported a potential SFTS case infected in Chiba Prefecture in 2017 (Taira et al., 2021).
While multiple human-to-human transmission cases of SFTSV have been reported in China and South Korea, Japan's first human-to-human transmission case was reported in March 2024. The case was a healthcare worker who had direct contact with bodily fluids from a severely ill patient infected with SFTSV (Seiji et al., 2024).
Analysis of national SFTS patients indicated that age, thrombocytopenia, coexisting malignancy, and the presence of tremor at the initial visit were significantly associated with poor prognosis in fatal cases (Kobayashi et al., 2020).
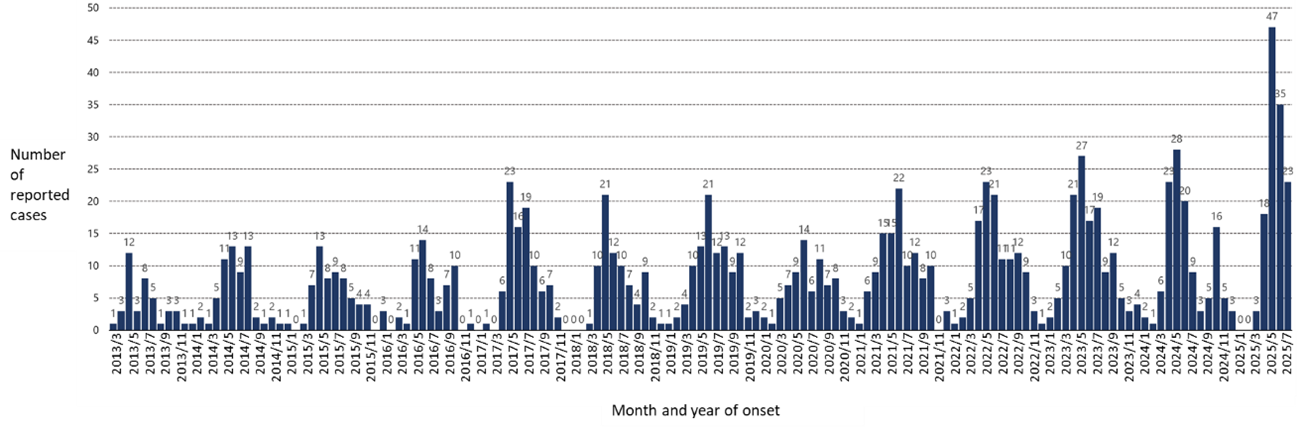 Figure 1. Month and year of onset for SFTS cases reported to the Infectious Disease Surveillance System (n=1,173, as of July 31, 2025)
Figure 1. Month and year of onset for SFTS cases reported to the Infectious Disease Surveillance System (n=1,173, as of July 31, 2025)
Table 1. Demographics on SFTS cases reported to the Infectious Disease Surveillance System (As of July 31, 2025)
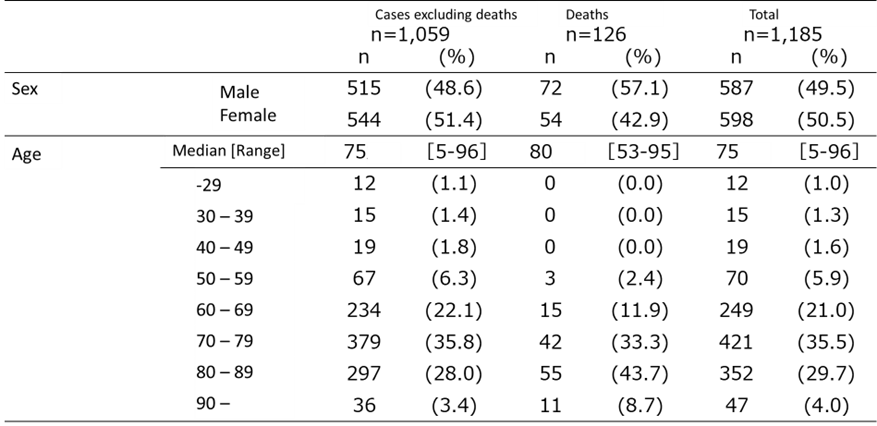
Figure 2. Reporting regions for SFTS cases (n=1,185, as of 31 July 2025)
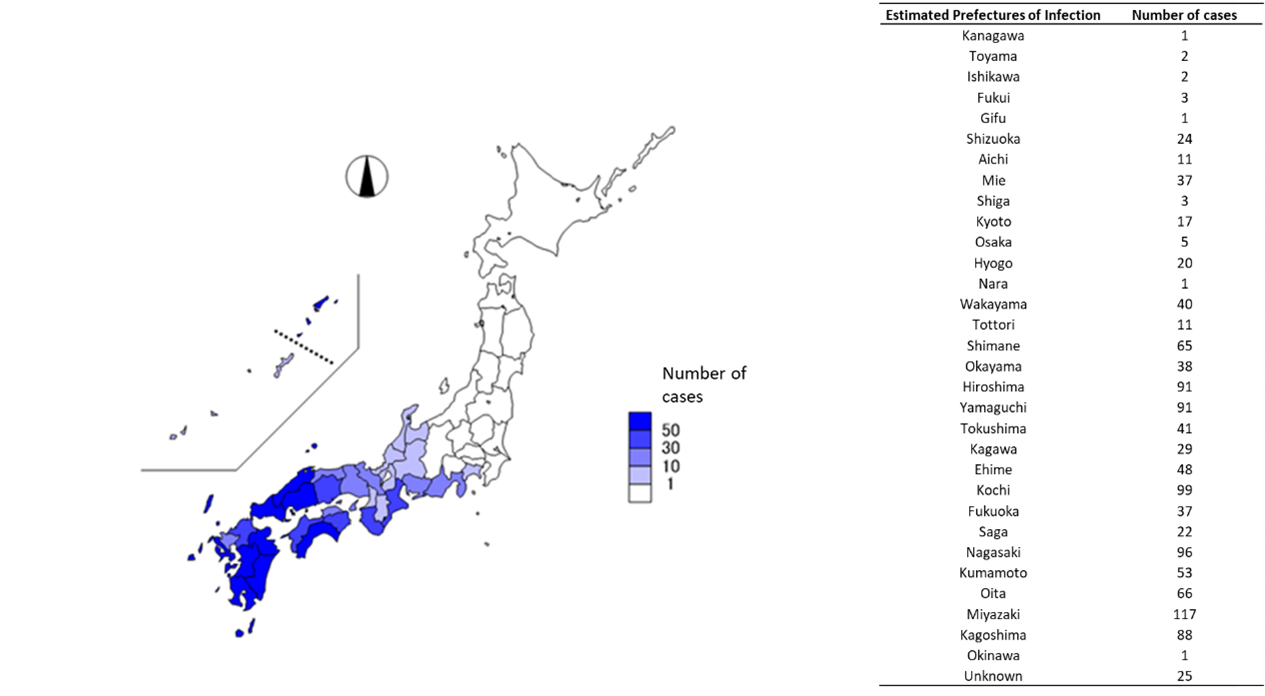
Figure 3. Estimated Region of Infection for Reported SFTS Cases (n=1,185, as of 31 July 2025)
2.Animal cases
In Japan, there is no legal reporting obligation for SFTS cases in animals. Domestic animal cases of SFTSV infection have been confirmed in a cat (April 2017), a dog (June 2017), and a cheetah (August 2017) (Pet al., 2022; Matsuno K. et al., 2018). Among these, the June 2017 dog case strongly suggested transmission to its owner. Since then, definitive diagnosis has been made through serum genetic testing when cats or dogs exhibit fever, leukopenia, thrombocytopenia, elevated liver enzymes, elevated CK, elevated T-Bil, jaundice, vomiting, or gastrointestinal symptoms. Additionally, elevated IgM antibodies detected by ELISA and increased IgG antibodies, or rise of viral neutralizing antibody titers in paired sera between acute and convalescent phases are used as supplementary diagnostic indicators.
Surveys of infection status in domestic animals conducted by the SFTS Diagnosis Network, organized with the cooperation of many researchers, reported 194 cat and 12 dog SFTS cases in Japan during 2024 (Figure 4). Most cases occurred in western Japan, though recent report emerged from Ibaraki Prefecture (Figure 5). The peak of cases in cats is from March to May, tending to be earlier than in humans (Figure 6). Cases also occur frequently in winter. Severe symptoms have been reported in cats, including fever, leukopenia, thrombocytopenia, and jaundice, with a reported CFR of 62.5% (Matsuu A. et al., 2019). Similar symptoms to those seen in cats and humans have also been observed in dogs (Ishijima K. et al., 2022). Each year, several instances of human infection involving pet owners or veterinary staff in contact with infected animals are reported (Oshima H. et al., 2022; Kirino Y. et al., 2022; Miyauchi A. et al., 2022; Kirino Y. et al., 2021; Tsuru M. et al., 2021; Kida K. et al., 2019, Tanaka et al., 2025). Therefore, strict infection control measures are essential when handling infected animals.
Wild animals often are infected through tick bites. The rise in infection rates rise among wildlife are reported to precede infection to human. Therefore, investigating antibody prevalence in wildlife may enable prediction of regional SFTS infection risk (Tatemoto K. et al., 2022a). Serological survey results in wildlife vary significantly by year and location; In western Japan, SFTSV antibody prevalence rates in wild boar and deer often exceeds 20%, while rates in Hokkaido and Tohoku region are below 20% (Table 2) (Tatemoto K. et al., 2022b, unpublished data).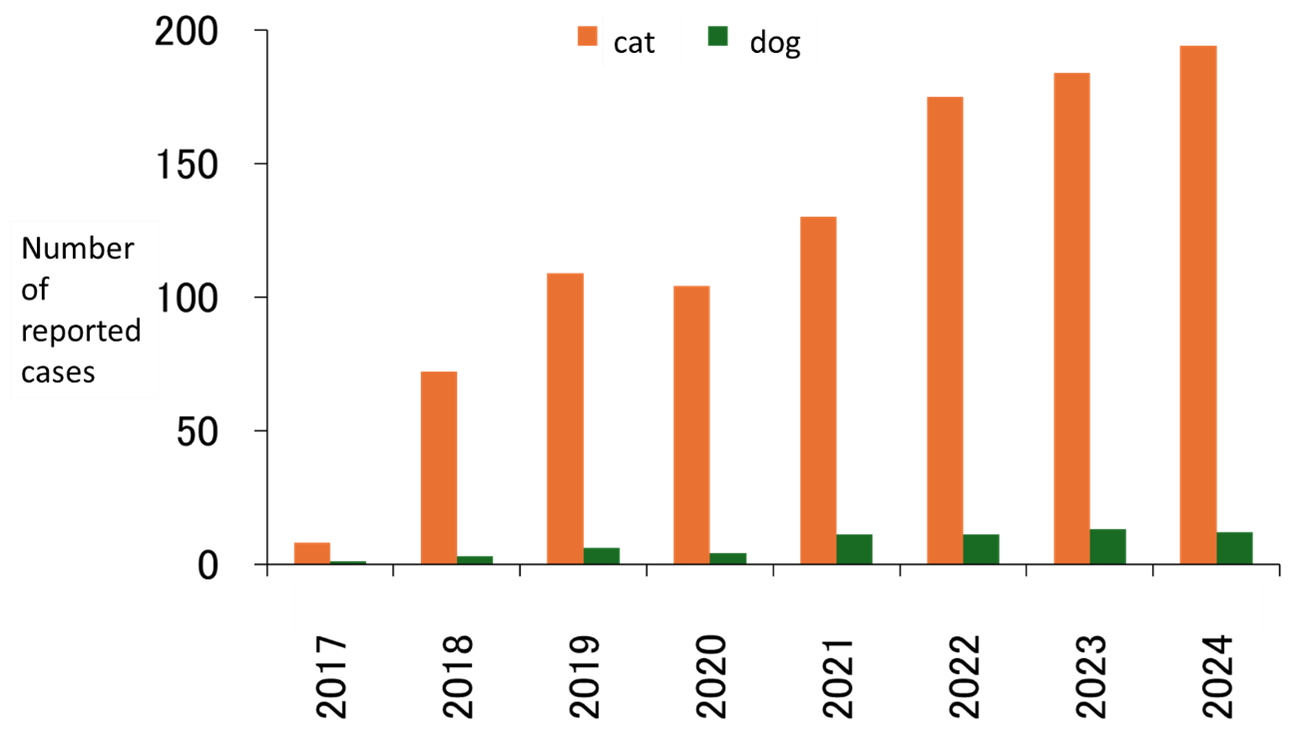 Figure 4. Annual number of SFTS cases in dogs and cats in Japan(2017–2024, as of 31 December 2024)
Figure 4. Annual number of SFTS cases in dogs and cats in Japan(2017–2024, as of 31 December 2024)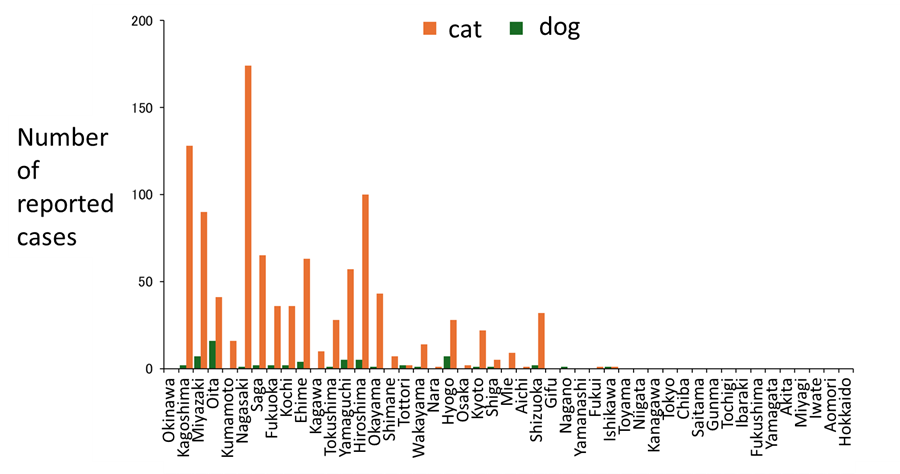
Figure 5. Number of SFTS cases in dogs and cats in Japan by region (2017–2024, as of 31 December 2024)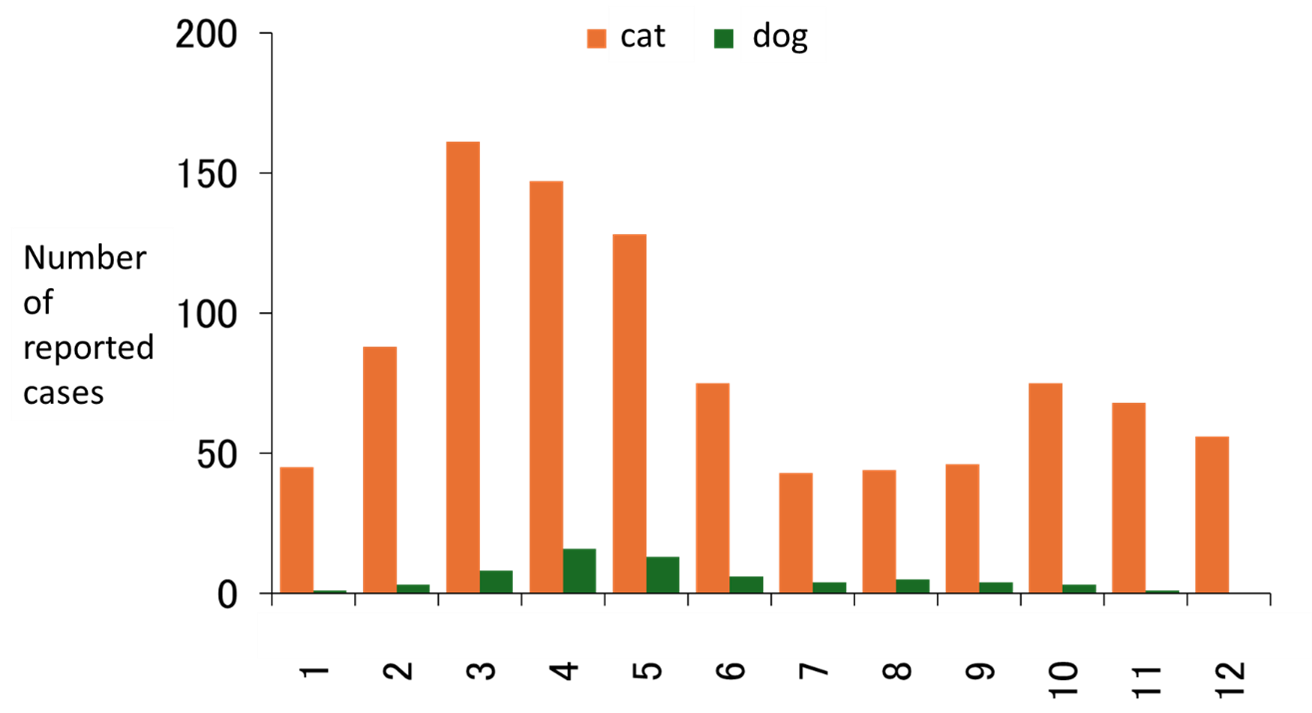
Figure 6. Monthly number of SFTS cases in dogs and cats in Japan(2017–2024, as of 31 December 2024)
Table 2. Prevalence of anti-SFTSV antibodies in wild deer and boar in Japan since 2007 (as of 30 March 2025)
|
Area of |
Deer |
Boar |
|||||
|
Number |
Number of |
Positive |
Number |
Number of |
Positive |
||
|
Hokkaido / |
25 |
0 |
0 |
||||
|
Hokkaido / |
53 |
1 |
2 |
8 |
0 |
0 |
|
|
Hokkaido / |
66 |
0 |
0 |
||||
|
Hokkaido / |
135 |
21 |
15.6 |
||||
|
Hokkaido / |
4 |
0 |
0 |
||||
|
Kanto A |
81 |
0 |
0 |
170 |
2 |
1.2 |
|
|
Kanto B |
189 |
0 |
0 |
46 |
0 |
0 |
|
|
Kanto C |
107 |
24 |
22.4 |
75 |
5 |
7 |
|
|
Kanto D |
37 |
2 |
5 |
||||
|
Chubu A |
171 |
15 |
8.8 |
||||
|
Chubu B |
200 |
4 |
2.0 |
||||
|
Chubu C |
513 |
7 |
1.4 |
144 |
4 |
2.8 |
|
|
Chubu D |
138 |
15 |
10.9 |
||||
|
Chubu E |
182 |
16 |
8.8 |
||||
|
Kansai A |
104 |
13 |
12.5 |
||||
|
Kansai B |
141 |
17 |
12.1 |
||||
|
Kansai C |
96 |
18 |
19 |
2 |
0 |
0 |
|
|
Kansai D |
155 |
40 |
25.8 |
||||
|
Kansai E |
668 |
261 |
39.1 |
824 |
459 |
55.7 |
|
|
Chugoku A |
42 |
6 |
14 |
||||
|
Chugoku B |
75 |
47 |
63 |
||||
|
Chugoku C |
37 |
24 |
65 |
||||
|
Chugoku D |
892 |
488 |
54.7 |
787 |
313 |
39.8 |
|
|
Shikoku A |
75 |
1 |
1 |
136 |
52 |
38.2 |
|
|
Shikoku B |
73 |
18 |
25 |
311 |
111 |
35.7 |
|
|
Shikoku C |
36 |
8 |
22 |
47 |
3 |
6 |
|
|
Kyushu/ |
36 |
3 |
8 |
||||
|
Kyushu/ |
30 |
22 |
73 |
47 |
3 |
6 |
|
|
Kyushu/ |
156 |
7 |
4.5 |
5 |
3 |
60 |
|
|
Kyushu/ |
64 |
11 |
17 |
182 |
130 |
71.4 |
|
|
Kyushu/ |
97 |
5 |
5 |
||||
|
Total |
4,399 |
1,073 |
24.4 |
3,062 |
1,117 |
36.5 |
|
6.Domestic countermeasures
The primary route of SFTSV transmission to humans is through the bite of infected ticks. However, infections have also been reported through bites from infected companion animals such as cats or dogs, as well as direct contact with bodily fluids of infected animals or with SFTS. Therefore, preventive measures against tick bites are essential, not only in western Japan where most SFTS cases in humans and animals are reported, but across the country. Additionally, when handling weakened companion animals who may develop SFTS, or SFTS patients, strict adherence to standard precautions and transmission-specific precautions is essential to avoid direct contact with bodily fluids.
To avoid tick bites, precautions are necessary when entering areas where ticks are prevalent, such as grassy areas and thickets, especially during spring through fall when ticks activity peaks and the risk of bites increases. It is crucial to minimize skin exposure by wearing long sleeves and long pants, tucking shirts into pants and pant into socks or boots, wearing hiking gaiters, avoiding sandals and wearing closed shoes that fully cover the feet, wearing a hat and gloves, and a towel around the neck. Additionally, light-colored clothing is recommended as to make ticks easier to visually detect. After outdoor activities, bathe and check for tick bites, paying particular attention to the neck, ears, armpits, groin, wrists, and behind the knees.
Furthermore, repellents containing DEET or picaridin as the main ingredient, sold as pharmaceuticals or quasi-drugs, are also effective for tick prevention. Only products that state “tick repellency” in their efficacy and effects section should be used, following the recommended dosage and instructions. The duration of repellent effectiveness is proportional to its concentration. For detailed tick prevention measures, refer to the educational material “Tick Prevention: What You Can Do Now” (Japan Institute for Health Security, 2025) developed by the Department of Medical Entomology.
Ticks typically insert their mouthparts into the skin of humans or animals firmly, and feed for extended periods from several days to over 10 days, however one may not notice the bite. When discovering a tick attached to the skin, it should be not removed forcefully, as it may cause mouthparts to remain embedded and pose a risk of infection, or force tick body fluids into the wound, increasing the risk of transmission. Medical evaluation at a healthcare facility, such as a dermatology clinic, is recommended for proper removal and wound disinfection. Additionally, after a tick bite, individuals should monitor their health condition for several weeks. If symptoms such as fever develop, prompt medical attention should be sought, and the clinician should be informed of the history of a tick bite.
Similarly, it is important to keep domesticated animals, particularly companion animals that frequently interact with humans, away from environments where ticks are common, such as grassy or wooded areas. If an animal has been in such an environment, visually inspect it for ticks. Regular use of tick preventatives (oral or topical) for companion animals is also effective to eliminate ticks that may have attached.
If an animal exhibits symptoms such as fever after a tick bite or exposure to environment where ticks are present, it is recommended to promptly seek veterinary care. During handling, protective measures such as wearing gloves and masks should be taken to prevent bites or contact with potentially infectious body fluids.
These tick prevention measures are effective not only against SFTS but also against other tick-borne infections such as Japanese spotted fever and Lyme disease.
Regarding treatment for SFTS, until recently, there were no antiviral drugs approved for STFS, limiting options to symptomatic therapy. However, on May 24, 2024, the Second Committee on Drugs of Pharmaceutical Affairs Council, MHLW, agreed on the addition of the indication for SFTS to favipiravir. This was officially approved on June 24 of the same year (Fujifilm Toyama Chemical Co., Ltd., 2024).
For detailed information on infection control measures and treatment for SFTS in medical institutions, refer to the “Guidelines for the Diagnosis and Treatment of Severe Fever with Thrombocytopenia Syndrome (SFTS) 2024 Edition” (Fiscal Year 2024 Ministry of Health, Labour and Welfare Science Research Grant, Research on Emerging and Re-emerging Infectious Diseases and Immunization, “Research on Establishing Clinical Response and Coordination Systems with Administrative Authorities in Preparation for Outbreaks of Class I Infectious Diseases”).
Additionally, for veterinary practitioners, the MHLW website provides a Q&A for specialists (https://www.mhlw.go.jp/bunya/kenkou/kekkaku-kansenshou19/sfts_qa.html), offering information on precautions and requesting tests when necessary.
7.Risk assessment
- SFTS is a tick-borne infectious disease reported in East and Southeast Asia, with the majority of human cases reported from China, South Korea, and Japan.
- In Japan, most of the estimated infection areas had primarily been in the western regions; however, human and animal cases is now also reported in the Kanto region and Hokkaido, suggesting a nationwide risk of infection.
- The risk of infection increases from spring to autumn, when ticks are most active. The infection risk is considered moderate for individuals engaging in outdoor activities during this period, while the risk in everyday setting is considered low.
- In addition to animal-to-human transmission reported both domestically and internationally, human-to-human transmission previously reported overseas has now also been reported in Japan. Therefore, the risk of infection is assessed moderate for healthcare and veterinary workers who come into contact with confirmed or suspected SFTS patients, their remains, animals, or animal carcasses.
8.Future Measures and Recommendations
- To prevent SFTSV infection through tick bites, individuals should take appropriate prevention measures when engaging in outdoor activities in grassy or wooded areas during high-risk seasons. Recommended measures include wearing long sleeves and long pants and applying repellents. These are also effective against other tick-borne diseases such as Japanese spotted fever.
- To prevent infection from animals or patients, appropriate infection control measures are necessary when handling confirmed or suspected SFTS patients, their remains, animals, or animal carcasses.
Related informations
- Japan Institute for Health Security. Severe Fever with Thrombocytopenia Syndrome (SFTS). (In Japanese)
- Japan Institute for Health Security. Summary and Evaluation of Infectious Disease Surveillance Information. Trends in SFTS Cases Reported through the Surveillance System for Infectious Diseases. (In Japanese)
- Japan Institute for Health Security. Pathogen Detection Manual: Severe Fever with Thrombocytopenia Syndrome (SFTS) 2nd Edition. (In Japanese)
- Japan Institute for Health Security. Tick Prevention: What You Can Do Now. (In Japanese)
- Ministry of Health, Labour and Welfare. Guidelines for the Diagnosis and Treatment of Severe Fever with Thrombocytopenia Syndrome (SFTS) 2024 Edition. https://www.mhlw.go.jp/content/10900000/001229138.pdf (In Japanese)
- Ministry of Health, Labour and Welfare. Management and Regulation of Specified Pathogens under the Infectious Diseases Control Law. https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/kenkou_iryou/kekkaku-kansenshou17/03.html (In Japanese)
- Ministry of Health, Labour and Welfare. Q&A on Severe Fever with Thrombocytopenia Syndrome (SFTS). https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/kenkou_iryou/kenkou/kekkaku-kansenshou19/sfts_qa.html (In Japanese)
References
- Chen Y, Jia B, Liu Y, Huang R, Chen J, Wu C. Risk factors associated with fatality of severe fever with thrombocytopenia syndrome: a meta-analysis. Oncotarget. 2017 Jul 11;8(51):89119-89129. doi: 10.18632/oncotarget.19163. PMID: 29179504; PMCID: PMC5687674.
- Denic S, Janbeih J, Nair S, Conca W, Tariq WU, Al-Salam S. Acute Thrombocytopenia, Leucopenia, and Multiorgan Dysfunction: The First Case of SFTS Bunyavirus outside China?. Case Rep Infect Dis. 2011;2011:204056. doi:10.1155/2011/204056.
- Hu J, Shi C, Li Z, et al. A cluster of cases of severe fever with thrombocytopenia syndrome bunyavirus infection in China, 1996: A retrospective serological study. PLoS Negl Trop Dis. 2018;12(6):e0006603. Published 2018 Jun 25. doi:10.1371/journal.pntd.0006603.
- Ishijima K, Tatemoto K, Park E, et al. Lethal Disease in Dogs Naturally Infected with Severe Fever with Thrombocytopenia Syndrome Virus. 2022;14(9):1963. Published 2022 Sep 4. doi:10.3390/v14091963.
- 2024 감염병 신고 현황 연보. https://dportal.kdca.go.kr/pot/bbs/BD_selectBbsList.do?q_bbsSn=1010&q_clsfNo=1.
- 疾病監視戦略担当官. 해외감염병발생동향제19호 Published 2024 May. https://dportal.kdca.go.kr/pot/bbs/BD_selectBbs.do?q_bbsSn=1009&q_bbsDocNo=20240516185737462&q_clsfNo=-1.
- Kida K, Matsuoka Y, Shimoda T, et al. A Case of Cat-to-Human Transmission of Severe Fever with Thrombocytopenia Syndrome Virus. Jpn J Infect Dis. 2019;72(5):356-358. doi:10.7883/yoken.JJID.2018.526.
- Kim KH, Yi J, Kim G, et al. Severe fever with thrombocytopenia syndrome, South Korea, 2012. Emerg Infect Dis. 2013;19(11):1892-1894. doi:10.3201/eid1911.130792.
- Kim KH, Lee MJ, Ko MK, Lee EY, Yi J. Severe Fever with Thrombocytopenia Syndrome Patients with Hemophagocytic Lymphohistiocytosis Retrospectively Identified in Korea, 2008-2013. J Korean Med Sci. 2018;33(50):e319. Published 2018 Nov 16. doi:10.3346/jkms.2018.33.e319.
- Kim YR, Yun Y, Bae SG, et al. Severe Fever with Thrombocytopenia Syndrome Virus Infection, South Korea, 2010. Emerg Infect Dis. 2018;24(11):2103-2105. doi:10.3201/eid2411.170756.
- Kirino Y, Ishijima K, Miura M, et al. Seroprevalence of Severe Fever with Thrombocytopenia Syndrome Virus in Small-Animal Veterinarians and Nurses in the Japanese Prefecture with the Highest Case Load. 2021;13(2):229. Published 2021 Feb 2. doi:10.3390/v13020229.
- Kirino Y, Yamanaka A, Ishijima K, Tatemoto K, Maeda K, Okabayashi T. Retrospective study on the possibility of an SFTS outbreak associated with undiagnosed febrile illness in veterinary professionals and a family with sick dogs in 2003. J Infect Chemother. 2022;28(6):753-756. doi:10.1016/j.jiac.2022.02.011.
- Kobayashi Y, Kato H, Yamagishi T, et al. Severe Fever with Thrombocytopenia Syndrome, Japan, 2013-2017. Emerg Infect Dis. 2020;26(4):692-699. doi:10.3201/eid2604.191011.
- Liu Y, Li Q, Hu W, et al. Person-to-person transmission of severe fever with thrombocytopenia syndrome virus. Vector Borne Zoonotic Dis. 2012;12(2):156-160. doi:10.1089/vbz.2011.0758.
- Liu, W., Lu, Q. B., Cui, N., Li, H., Wang, L. Y., Liu, K., Yang, Z. D., Wang, B. J., Wang, H. Y., Zhang, Y. Y., Zhuang, L., Hu, C. Y., Yuan, C., Fan, X. J., Wang, Z., Zhang, L., Zhang, X. A., Walker, D. H., & Cao, W. C. (2013). Case-fatality ratio and effectiveness of ribavirin therapy among hospitalized patients in china who had severe fever with thrombocytopenia syndrome. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America, 57(9), 1292–1299. https://doi.org/10.1093/cid/cit530.
- Matsuno K, Nonoue N, Noda A, et al. Fatal Tickborne Phlebovirus Infection in Captive Cheetahs, Japan. Emerg Infect Dis. 2018;24(9):1726-1729. doi:10.3201/eid2409.171667.
- Matsuu A, Momoi Y, Nishiguchi A, et al. Natural severe fever with thrombocytopenia syndrome virus infection in domestic cats in Japan. Vet Microbiol. 2019;236:108346. doi:10.1016/j.vetmic.2019.06.019.
- Miyauchi A, Sada KE, Yamamoto H, et al. Suspected Transmission of Severe Fever with Thrombocytopenia Syndrome Virus from a Cat to a Veterinarian by a Single Contact: A Case Report. Viruses. 2022;14(2):223. Published 2022 Jan 24. doi:10.3390/v14020223.
- Oshima H, Okumura H, Maeda K, Ishijima K, Yoshikawa T, Kurosu T, Fukushi S, Shimojima M, Saijo M. A Patient with Severe Fever with Thrombocytopenia Syndrome (SFTS) Infected from a Sick Dog with SFTSV Infection. Jpn J Infect Dis. 2022 Jul 22;75(4):423-426. doi: 10.7883/yoken.JJID.2021.796. Epub 2022 Feb 28. PMID: 35228501.
- Peng SH, Yang SL, Tang SE, et al. Human Case of Severe Fever with Thrombocytopenia Syndrome Virus Infection, Taiwan, 2019. Emerg Infect Dis. 2020;26(7):1612-1614. doi:10.3201/eid2607.200104.
- Rattanakomol P, Khongwichit S, Linsuwanon P, Lee KH, Vongpunsawad S, Poovorawan Y. Severe Fever with Thrombocytopenia Syndrome Virus Infection, Thailand, 2019-2020. Emerg Infect Dis. 2022;28(12):2572-2574. doi:10.3201/eid2812.221183.
- Tatemoto K, Ishijima K, Kuroda Y, et al. Roles of raccoons in the transmission cycle of severe fever with thrombocytopenia syndrome virus. J Vet Med Sci. 2022;84(7):982-991. doi:10.1292/jvms.22-0236. 2022
- Tatemoto K, Virhuez Mendoza M, Ishijima K, Kuroda Y, Inoue Y, Taira M, Kuwata R, Takano A, Morikawa S, Shimoda H. Risk assessment of infection with severe fever with thrombocytopenia syndrome virus based on a 10-year serosurveillance in Yamaguchi Prefecture. J Vet Med Sci. 2022;84(8):1142-1145. doi: 10.1292/jvms.22-0255. 2022b.
- Tran XC, Yun Y, Van An L, et al. Endemic Severe Fever with Thrombocytopenia Syndrome, Vietnam. Emerg Infect Dis. 2019;25(5):1029-1031. doi:10.3201/eid2505.181463.
- Tsuru M, Suzuki T, Murakami T, et al. Pathological Characteristics of a Patient with Severe Fever with Thrombocytopenia Syndrome (SFTS) Infected with SFTSV through a Sick Cat's Bite. 2021;13(2):204. Published 2021 Jan 29. doi:10.3390/v13020204.
- Win AM, Nguyen YTH, Kim Y, et al. Genotypic Heterogeneity of Orientia tsutsugamushi in Scrub Typhus Patients and Thrombocytopenia Syndrome Co-infection, Myanmar. Emerg Infect Dis. 2020;26(8):1878-1881. doi:10.3201/eid2608.200135.
- Xu B, Liu L, Huang X, et al. Metagenomic analysis of fever, thrombocytopenia and leukopenia syndrome (FTLS) in Henan Province, China: discovery of a new bunyavirus. PLoS Pathog. 2011;7(11):e1002369. doi:10.1371/journal.ppat.1002369.
- Yu XJ, Liang MF, Zhang SY, et al. Fever with thrombocytopenia associated with a novel bunyavirus in China. N Engl J Med. 2011;364(16):1523-1532. doi:10.1056/NEJMoa1010095.
- Yue Y, Ren D, Lun X. Epidemic characteristics of fatal cases of severe fever with thrombocytopenia syndrome in China from 2010 to 2023. J Trop Dis Parasitol. 2024;22(5):257–261. doi:10.20199/j.issn.1672-2302.2024.05.001.
- Yue Y, Ren D, Lu L. Epidemiological characteristics of severe fever with thrombocytopenia syndrome in China, 2010–2021. Dis Surveill. 2024;39(7):824–830. doi:10.3784/jbjc.202307030309.
- Zhang Q, Liu W, Wang W, et al. Analysis of spatial-temporal distribution characteristics and natural infection status of SFTS cases in Hefei from 2015 to 2021. Environ Health Prev Med. 2023;28:70. doi:10.1265/ehpm.23-00149.
- Chinese CDC, Technical guidelines for prevention and control of severe fever with thrombocytopenia syndrome (SFTS), 2024 edition. https://www.cqcdc.org/uploads/ueditor/file/20241008/1728371799916003.pdf
- S Kiyotoki et al, IASR. 2024;45:62–64. (In Japanese)
- Shi H et al, Epidemic Status and Risk Assessment of Severe Fever with Thrombocytopenia Syndrome in Taiwan. Taiwan Epidemiology bulletin, 39(4):46-55. https://www.cdc.gov.tw/En/File/Get/NfgCSp3mwh1oRFF8pY2l_Q. (In Chinese)
- Ibaraki Prefecture. Occurrence of the first SFTS case in Ibaraki Prefecture. 2024. https://www.pref.ibaraki.jp/hokenfukushi/yobo/kiki/yobo/kansen/idwr/press/documents/0807.pdf. (In Japanese)
- Sapporo City. Occurrence of the first SFTS case in Hokkaido (August 7, 2025).
- https://www.city.sapporo.jp/hokenjo/f1kansen/documents/20250807press_2.pdf. (In Japanese)
- M Taira et al, IASR. 2021;42:150-152: 2021年7月号. https://id-info.jihs.go.jp/niid/ja/sfts/sfts-iasrs/10449-497p02.html. (In Japanese)
- H Tanaka et al, IASR. 2024;46:165-167. (In Japanese)
- Fujifilm Toyama Chemical Co., Ltd. Notice on additional indication approval of antiviral drug “Avigan® tablets.” Published 2024 Jun 24. https://www.fujifilm.com/fftc/ja/news/332. (In Japanese)

